The COVID-19 pandemic has accelerated the use of remote interactions between clinical trial investigators and patients and the adoption of new digital technologies. This has promoted the decentralization of the SCAC, with all the benefits that this entails. MultiHealth explains.
PMCF (Post Market Clinical Follow-up) is a continuous process of updating clinical evaluation. The manufacturer collects clinical data resulting from the use of a medical device (MD) in real life and proactively evaluates it to confirm performance and safety claims throughout the expected lifetime of the MD.
Decentralization offers the opportunity, thanks to digital tools, to rethink clinical investigation by bringing it closer to the source of data, namely the patient. The social distancing imposed by lockdown has significantly reinforced this trend.
The central role of digital tools
Decentralization involves dematerialization, i.e., the use of digital technologies by manufacturers, healthcare professionals, and patients alike.
Teleconsultation has been a great help during the Covid-19 crisis, with its use increasing due to health conditions. This method of consultation has now become commonplace for healthcare professionals and patients. These video consultations not only allow close contact with the patient to be maintained, but also enable remote assessment of whether patients are using a device correctly, which is of additional interest in the context of clinical investigations specific to a medical device.
The number of connected medical devices has multiplied in recent years. Integrated into remote monitoring platforms, they enable real-time reporting of patient data such as heart rate, sleep quality, and level of physical activity. In addition to these MDs, there are connected health devices such as connected scales and blood pressure monitors. Once these devices have proven their reliability, they can be included in the post-marketing clinical follow-up of medical devices, including for the purpose of collecting data related to prevention, risk reduction, or the impact on the progression of chronic pathologi
.
Digitization involves the digitization of forms used in clinical investigations, a practice that is already widespread,
whether it involves:
- obtain patient consent (e-consent),
- collect information entered directly by the patient (e-PRO, Patient Reported Outcome)
- or compile information collected by the medical team (e-CRF, Case Report Form) including demographic and clinical data, etc.
Finally, an essential component of decentralizing clinical investigations is remote source data verification (rSDV). As a general rule, monitoring ensures the reliability of the data collected and therefore enhances the quality of the study. In France, in April 2021, the CNIL published provisional recommendations concerning the remote quality control of source data.
Since December 31, 2021, these recommendations are no longer in effect. To date, there is no reference framework or Reference Methodology (MR) adapted to digitalized clinical trials, suggesting that it is no longer possible to use this remote monitoring method. However, rSDV is still relevant and more so than ever: it is carried out via a secure, validated platform dedicated to this purpose and authorized by the CNIL for each study. The absence of an MR means that a specific CNIL submission process must be anticipated, in the same way as the CNRIPH submission.
Involve local healthcare providers
Paramedical professionals contribute locally to patient care and to improving patients' conditions and quality of life. Although they are currently underrepresented in the design of clinical investigations, they play a key role in the patient journey. These professionals include:
- healthcare professionals: nurses, nursing assistants, midwives,
pediatric nurses, etc. - rehabilitation professionals: psychomotor therapists, massage therapists, dieticians, nutritionists, etc.
- orthopedic professions: opticians, orthoptists, audiologists, speech therapists, chiropodists, podiatrists, etc.
Training these mobile investigative teams in Good Clinical Practices (ISO 14155) and the regulatory context of medical devices would enable the deployment of efficient SCACs. Although these professions are currently in low demand, they represent an important lever for collecting quality data and maintaining the long-term commitment of patients participating in clinical follow-ups, which can sometimes be lengthy.
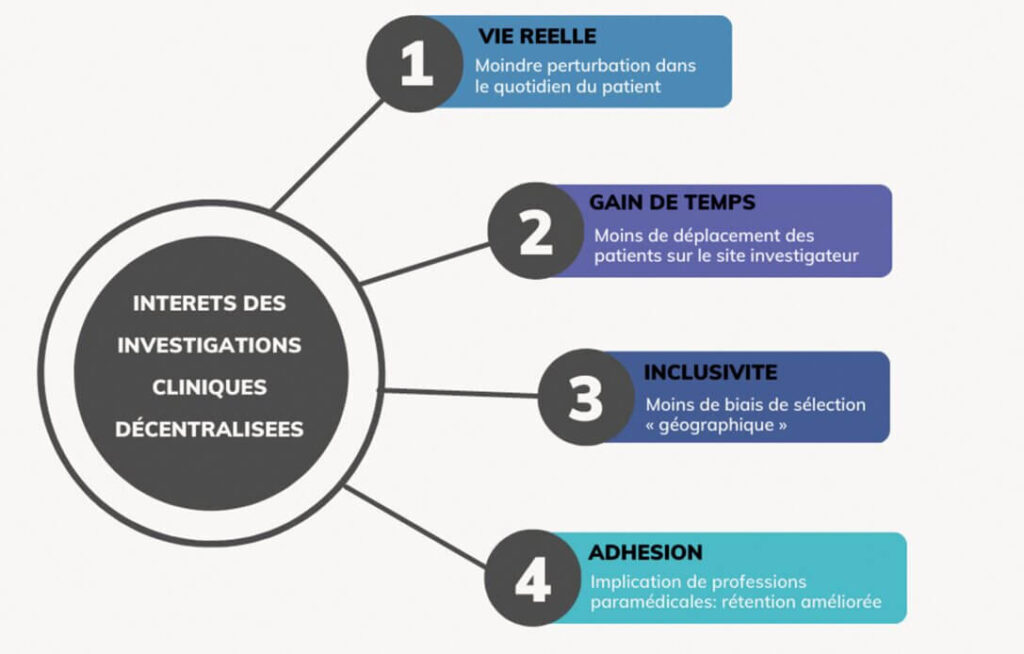
The benefits of decentralization
Decentralization is rarely considered in a 100% mode. Studies conducted with the support of digital technologies and tools are in fact most often hybrid, with limited visits to the investigator site combined with remote communication with the patient. In this context, there are multiple benefits, as shown in the figure above.
Reflection of real life: the adoption of decentralized methods helps to collect data outside the clinical environment, with less disruption to the patient's daily life and without interfering with normal usage patterns. These more "discreet" collection methods better reflect real-life post-marketing situations.- Time savings: collecting data via decentralized clinical investigations reduces the duration of investigations, as patients no longer need to travel to the investigation site to provide information.
- Inclusiveness: Health authorities and notified bodies require the most comprehensive representation possible of "beneficiary" patients. By conducting investigations as close as possible to patients, particularly through the use of digital technologies, "geographical" selection bias is limited and the diversity of participating patients is increased.
- Adherence: involving paramedical professionals who are closely involved in patients' daily lives helps to strengthen patient adherence to the study and ensures that a larger volume of data is collected over time. This improved retention also guarantees representativeness.
The decentralization of clinical investigations makes it possible to open up SCACs by reducing dependence on traditional research infrastructures specializing in data collection. However, there are MDs or IVD MDs (in vitro diagnostic medical devices) whose use in practice depends solely on a highly qualified or trained clinical expert. For example, long-term monitoring of devices related to diagnosis or certain procedures, such as the use of scanners or catheters, is not eligible for decentralization.
In conclusion, the methodology of decentralized clinical trials accelerated by the Covid-19 pandemic now represents a fundamental change in the way clinical evaluations are conducted. These changes in practice benefit the clinical follow-up of medical devices after they have been placed on the market. This requires training stakeholders—including paramedical professions—in clinical investigations and adapting service models to facilitate the use of digital solutions and their validation by regulatory authorities.
Decentralization makes it possible to collect real-life data from a more representative patient population. Agility in the service of diversity.
Frequently Asked Questions
Need assistance?
Are you planning a SCAC or a decentralized clinical investigation? CLINACT, a MultiHealth Group entity, supports you in the design, remote monitoring (rSDV), and regulatory submission (CNRIPH, CNIL) of your post-marketing studies.



